 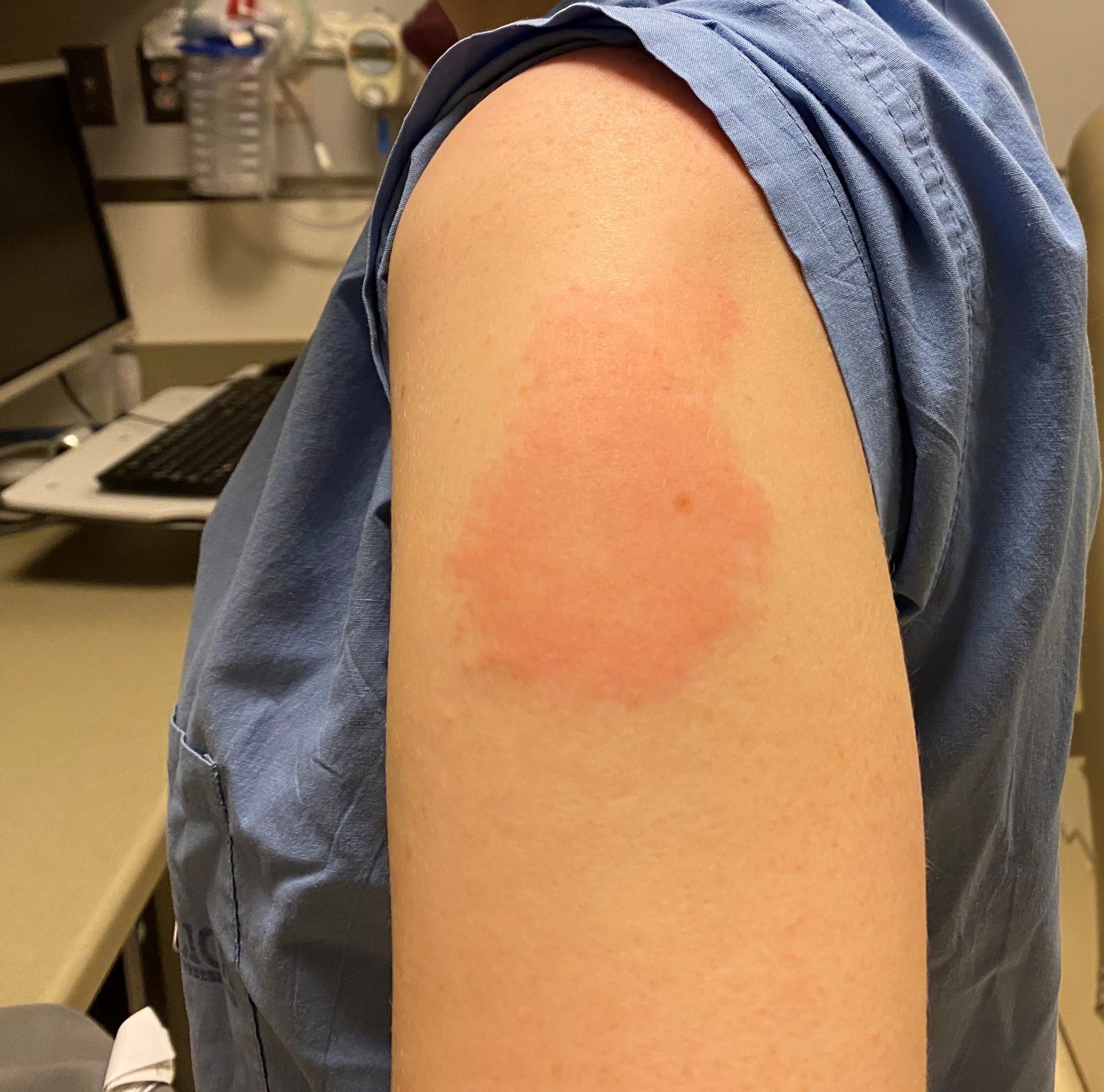
Frequency of the most common manifestations of local injection-site reactions, by dose, were swelling (18% and 15%), erythema (18% and 20%), and pain (24% and 18%). Local injection-site reactions occurred after 24% and 25% of first and second doses, respectively. Delayed large local reactions occurred after 15% of first doses and after 18% of second doses. Frequently found side effects include: Mild side. The Pfizer subgroup represented a smaller sample size (34 first-dose reports and 40 second-dose reports), but the pattern of skin reactions was similar to that observed with the Moderna vaccine. However, severe allergic reactions are often reported to develop within 15 minutes after vaccine is given. Onset is typically around one week after vaccination. As with any medicine, there is a very remote chance of a vaccine causing a severe allergic reaction, other serious injury, or death. If these problems occur, they usually begin soon after vaccination and are mild and short-lived. No other type of reaction occurred in more than 1%-2% of patients after either dose. COVID arm is a delayed hypersensitivity reaction that occurs mostly from the Moderna vaccine. Vomiting, muscle aches, fever, sore throat, and cough are other possible side effects. Erythromelalgia occurred after 5.9% of second doses, and cosmetic filler reactions occurred after 4.9% of second doses. 
Measles-like morbilliform rash occurred after the first dose of Moderna vaccine in 4.1% of patients and after the second dose in 6.9%. In some cases, the Takeda/Moderna vaccine caused delayed skin reactions at the. An additional 2% of patients developed urticaria within the first 24 hours (all after the second dose). In most cases, the rash arose more than 24 hours after dosing (4.8% after the first dose, 4.9% after the second). 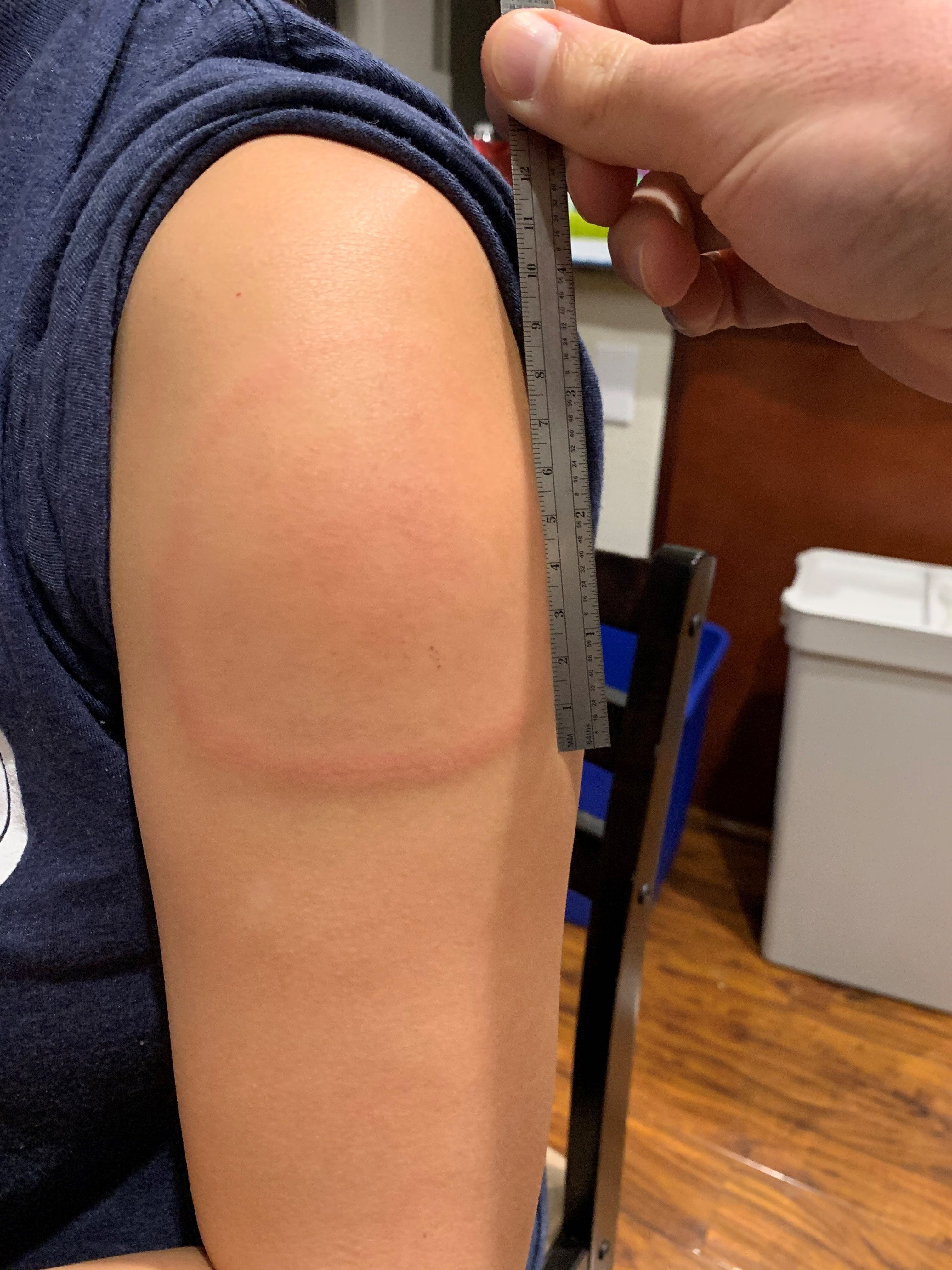
Urticaria was the next most common type of skin reaction associated with the Moderna vaccine. Registry participants collected information on the type and timing of vaccine doses and the morphology, timing, duration, and treatment of reactions. "Understandably, the clinical trials were focused on their major endpoints and didn't really provide a lot of detail on the skin reactions seen in patients."Įstablished in March 2020 to accumulate information on cutaneous manifestations of COVID-19, the registry expanded in December to include vaccine-related skin reactions, shortly after the FDA issued the first emergency use authorizations (EUAs) for the vaccines. "We have been involved with following skin reactions to COVID-19 vaccines, but the paper is pretty novel in that it covers a whole range of reactions that had not been reported from vaccine clinical trials," registry principal investigator Esther Freeman, MD, PhD, of Massachusetts General Hospital in Boston, told MedPage Today. 
They were discussed during the recent AAD virtual meeting and published simultaneously in the Journal of the American Academy of Dermatology. PMCID: PMC9895865.The findings came from the COVID-19 Dermatology Registry, a joint effort of the American Academy of Dermatology (AAD) and the International League of Dermatologic Societies. Delayed Cutaneous Reactions Following COVID-19 Vaccinations – A Military Cohort Analysis. Of these reaction reports, the Moderna vaccine was found to have the highest incidence of delayed cutaneous reactions, at a rate of 84.7%, over Johnson & Johnson and over Pfizer. The team noted that delayed cutaneous reactions were only reported in COVID-19 vaccine reports with "rash" or "hives," with a prevalence of 25.5%. Overall, the investigators’ results indicated that 2.2% of COVID-19 vaccine reports and 7.6% of Influenza vaccine reports had "rash" or "hives" mentioned.ĭespite this fact, the research team found no significant difference in the frequency of reported cutaneous reactions between COVID-19 and influenza vaccines. They reviewed reports that included "rash" or "hives" to determine when cutaneous symptoms occurred following vaccination. The researchers used a Vaccine Adverse Event Reporting System (VAERS) database specific to those who were eligible for military healthcare and analyzed adverse reaction reports in the period between December of 2020 and July of 2022 after both COVID-19 and influenza vaccinations. This study aims to investigate whether delayed cutaneous reactions are more frequently reported following COVID-19 vaccines compared to other available vaccines such as influenza. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
